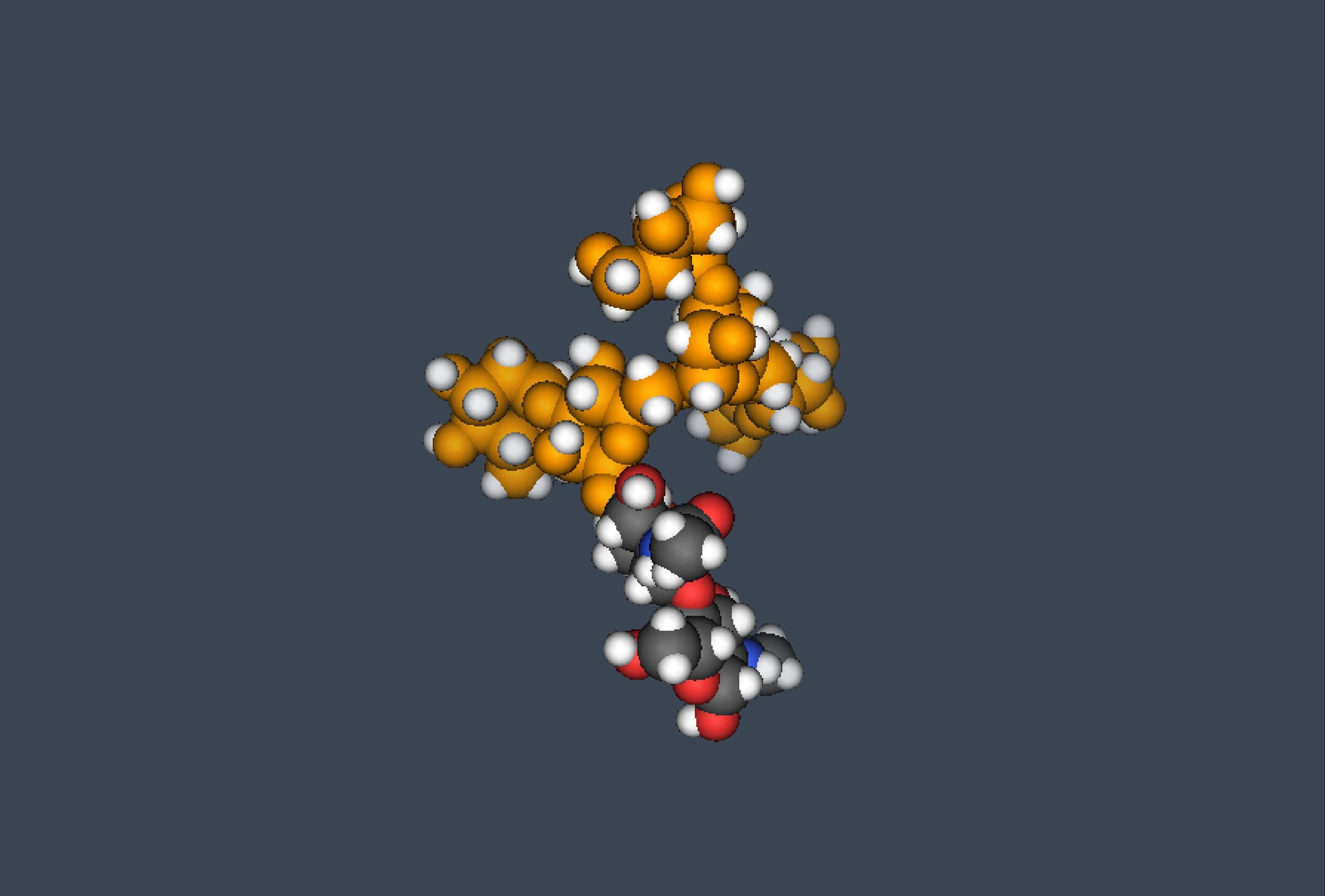
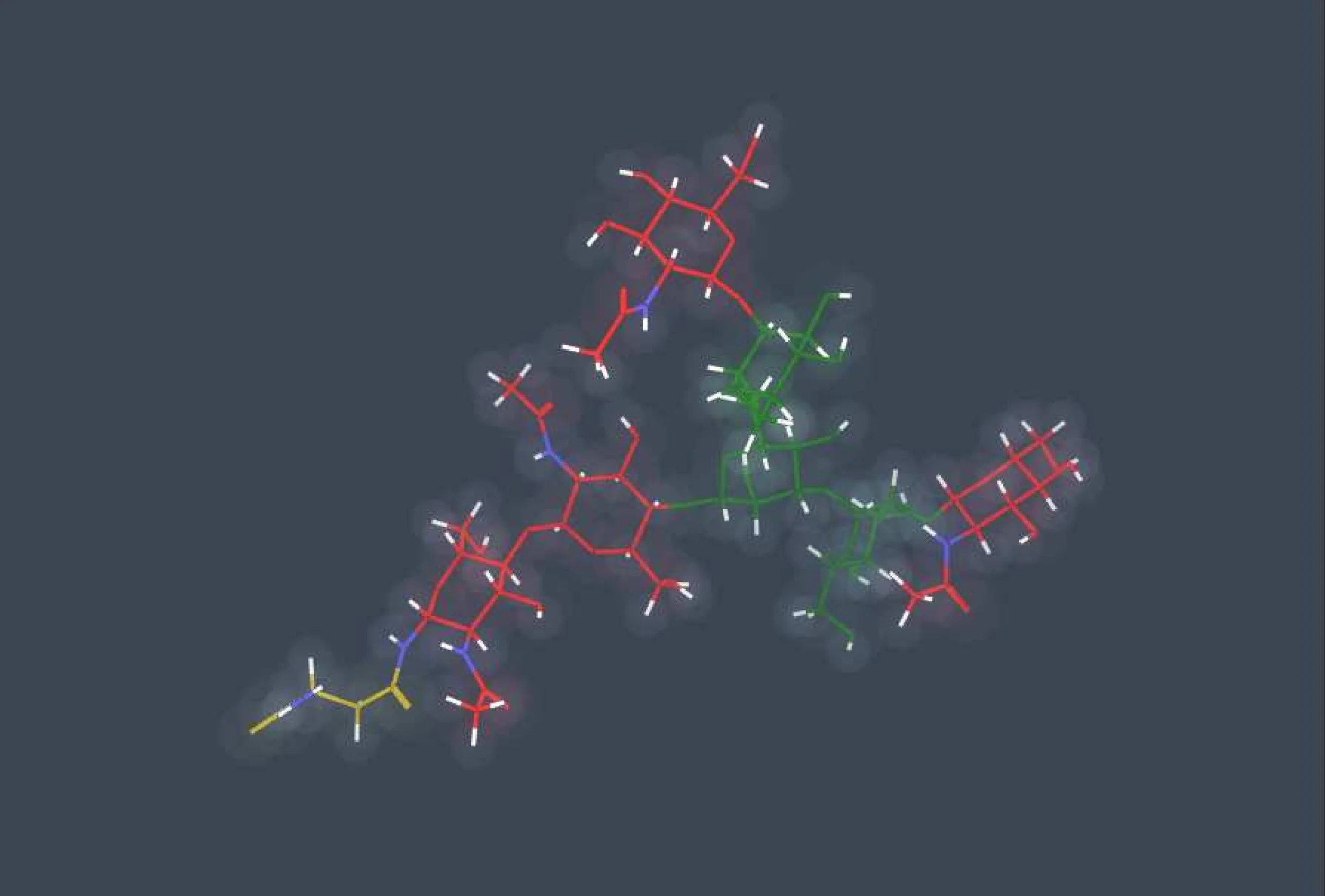
N-linked glycosylation
Glycomics #60_N-linked glycosylation (high mannose)
Glycomics #61_N-linked glycosylation (high mannose)
Glycomics #62_N-linked glycosylation (high mannose)
Glycomics #63_N-linked glycosylation (complex)
Glycomics #64_N-linked glycosylation (complex)
Glycomics #65_N-linked glycosylation (complex)
Glycomics #66-N-linked glycosylation (hybrid)
Glycomics #67_N-linked glycosylation (hybrid)
N-linked glycosylation
N-linked glycosylation-High Mannose
N-linked glycosylation, is the attachment of a glycan consisting of several sugar molecules to a nitrogen atom (the amide nitrogen of an asparagine (Asn) residue of a protein). This type of linkage is important for both the structure and function of many eukaryotic proteins. The N-linked glycosylation process occurs in eukaryotes and widely in archaea, but very rarely in bacteria. The nature of N-linked glycans attached to a glycoprotein is determined by the protein and the cell in which it is expressed. It also varies across species. Different species synthesize different types of N-linked glycans. The major fractions consisted of "high-mannose type" oligosaccharides containing five to nine mannose residues.
N-linked glycosylation-Complex
N-linked glycosylation, is the attachment of a glycan consisting of several sugar molecules to a nitrogen atom (the amide nitrogen of an asparagine (Asn) residue of a protein). This type of linkage is important for both the structure and function of many eukaryotic proteins. The N-linked glycosylation process occurs in eukaryotes and widely in archaea, but very rarely in bacteria. The nature of N-linked glycans attached to a glycoprotein is determined by the protein and the cell in which it is expressed. It also varies across species. Different species synthesize different types of N-linked glycans. N-linked glycosylation with Bi- and tri-antennary branches with the ubiquitous building block Galβ1-4GlcNAc, are called complex.
N-linked glycosylation-hybrid
N-linked glycosylation, is the attachment of a glycan consisting of several sugar molecules to a nitrogen atom (the amide nitrogen of an asparagine (Asn) residue of a protein). This type of linkage is important for both the structure and function of many eukaryotic proteins. The N-linked glycosylation process occurs in eukaryotes and widely in archaea, but very rarely in bacteria. The nature of N-linked glycans attached to a glycoprotein is determined by the protein and the cell in which it is expressed. It also varies across species. Different species synthesize different types of N-linked glycans.N-linked glycosylation with Bi- and tri-antennary branches with the ubiquitous building block Galβ1-4GlcNAc, and mannose residues are called hybrid.