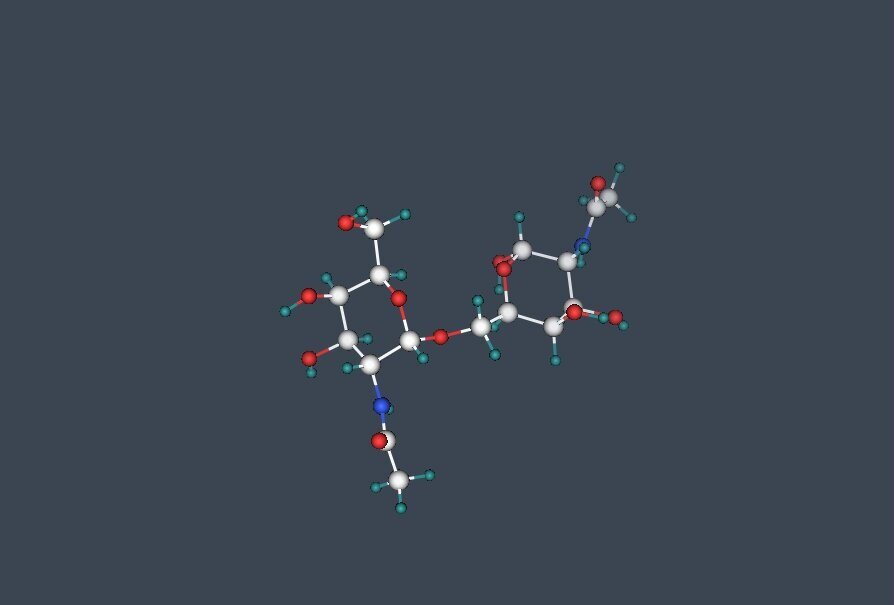
O-linked glycosylation
Glycomics #68_O-linked glycosylation (core 0)
Glycomics #69_O-linked glycosylation (core 1)
Glycomics #70_O-linked glycosylation (core 2)
Glycomics #71_O-linked glycosylation (core 3)
Glycomics #72_O-linked glycosylation (core 4)
Glycomics #73_O-linked glycosylation (core 5)
Glycomics #74_O-linked glycosylation (core 7)
Glycomics #75_Lewis a antigen
Glycomics #76_Lewis a antigen
Glycomics #77_Lewis b antigen
Glycomics #78_Lewis b antigen
Glycomics #79_Lewis c antigen
Glycomics #80_Lewis c antigen
Glycomics #88_sialyl Lewis a antigen
Glycomics #89_sialyl Lewis a antigen
Glycomics #90_sialyl Lewis a antigen
Glycomics #91_sialyl Lewis c antigen
Glycomics #92_sialyl Lewis c antigen
O-linked glycosylation
O-linked glycans are a common covalent modification of serine and threonine residues of mammalian glycoproteins. The most common type of O-linked glycans found on secreted mammalian glycoproteins and mucins (proteins conjugated to carbohydrate) is from the addition of N-acytylgalactosamine (GalNac) to serine or threonine residues. The Core 1 structure is generated by the addition of galactose b1-3 to this GalNAc. The Core 2 structure is generated by the further addition of N-acetyl-glucosamine b1-6 to the N-acetyl-galactosamine on the Core 1 structure. In total eight different O-linked glycans core structures have been identified and all of these can be further elongated by the addition of a number of monosaccharides including sialic acids.
All Glycomics 3D model were generated using SGI (Silicon Graphics) Octane IRIX workstation in 2002.